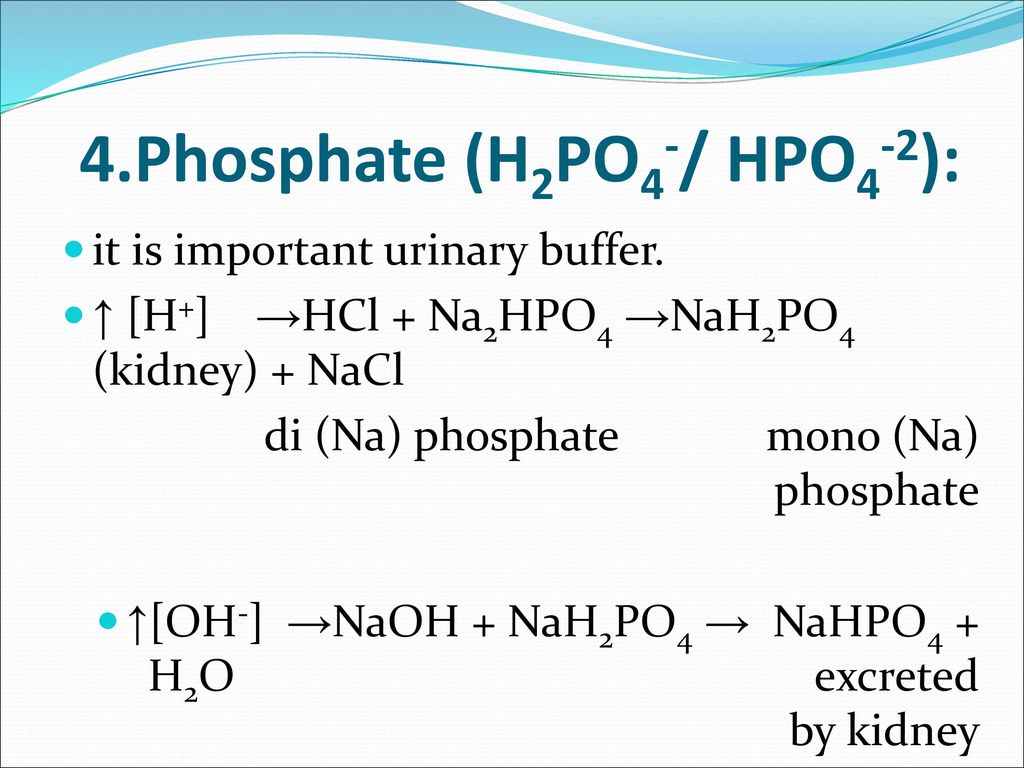
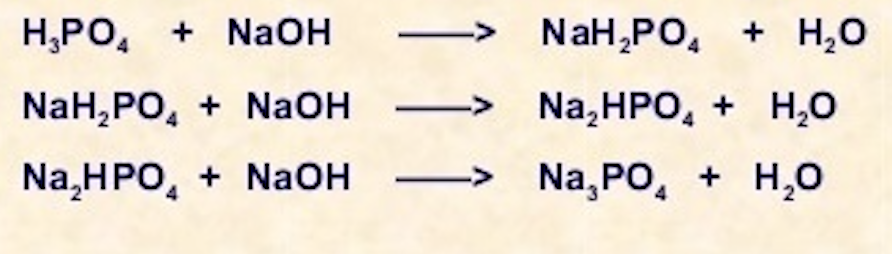
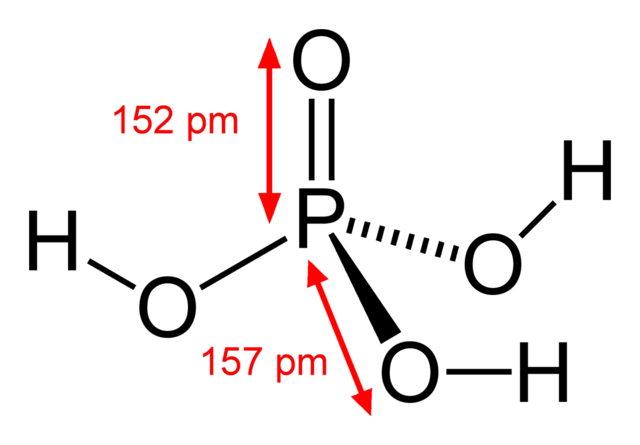
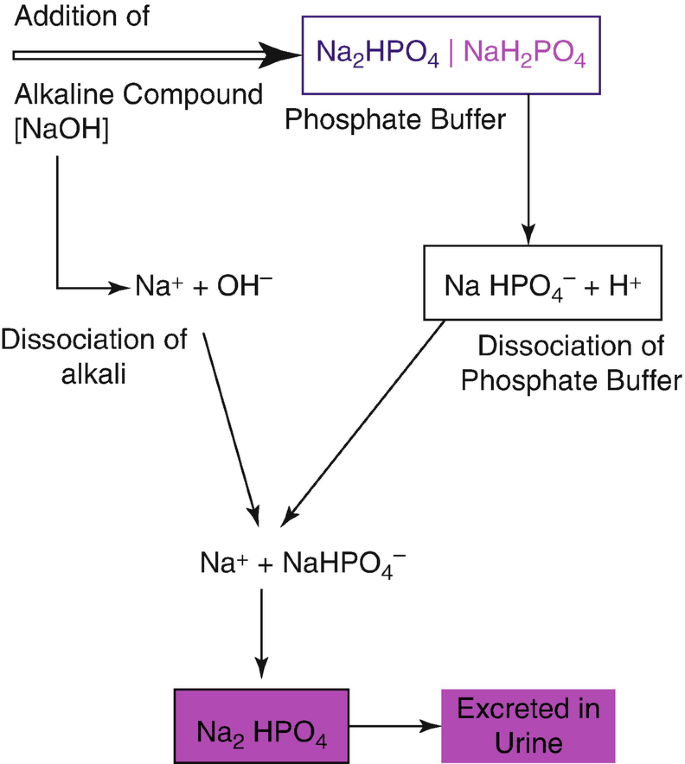
The Biological bulletin. Biology; Zoology; Biology; Marine Biology. 92 B. VERACHTERT AND A. DE LOOF Conway, 1941). NaCl 21.9 mM, NaH:PO4 1 mM NaHCO3 10 mM. MgCl2 0.12 mM. CaCl2 0.37

Phosphate Disodique Nahpo4 7758-79-4 Dsp - Buy Nahpo4 7758-79-4 Dsp,Disodique Phosphate Nahpo4 7758-79-4 Dsp Product on Alibaba.com

Dihydrate de phosphate de sodium dibasique, conforme aux normes analytiques de BP, Ph. Eur., 98,5 à 101 % (calc. à la substance séchée), Honeywell Fluka | Fisher Scientific
![Fluorescent Carbon Dots as Nanosensors for Monitoring and Imaging Fe3+ and [ HPO4]2– Ions | ACS Applied Nano Materials Fluorescent Carbon Dots as Nanosensors for Monitoring and Imaging Fe3+ and [ HPO4]2– Ions | ACS Applied Nano Materials](https://pubs.acs.org/cms/10.1021/acsanm.0c02515/asset/images/medium/an0c02515_0007.gif)
Fluorescent Carbon Dots as Nanosensors for Monitoring and Imaging Fe3+ and [ HPO4]2– Ions | ACS Applied Nano Materials

Corrosion rates obtained in 3.0% (NaCl + x% Na 2 HPO4) or in 3.0% (N/S... | Download Scientific Diagram

Phosphate Disodique Nahpo4 7758-79-4 Dsp - Buy Nahpo4 7758-79-4 Dsp,Disodique Phosphate Nahpo4 7758-79-4 Dsp Product on Alibaba.com

a) Crystal structure of Na2Fe(C2O4)(HPO4) viewed along the a-axis and... | Download Scientific Diagram
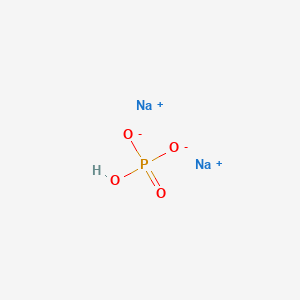
Sodium phosphate (NaHPO4) - Uses, DMF, Dossier, Manufacturer, Supplier, Licensing, Distributer, Prices, News, GMP

Vidéo de question : Déterminer quels produits chimiques fourniront les éléments importants pour la croissance des plantes | Nagwa
Prediction of Sodium Substitution Sites in Octacalcium Phosphate: The Relationships of Ionic Pair Ratios in Reacting Solutions
![Na(H3NCH2CH2NH3)0.5[Co(C2O4)(HPO4)]: A novel phosphoxalate open-framework compound incorporating both an alkali cation and an organic template in the structural tunnels - ScienceDirect Na(H3NCH2CH2NH3)0.5[Co(C2O4)(HPO4)]: A novel phosphoxalate open-framework compound incorporating both an alkali cation and an organic template in the structural tunnels - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022459607001934-fx1.jpg)
Na(H3NCH2CH2NH3)0.5[Co(C2O4)(HPO4)]: A novel phosphoxalate open-framework compound incorporating both an alkali cation and an organic template in the structural tunnels - ScienceDirect

SOLVED: What other combination Of phosphoric acid and its salts could be mixed to prepare this buffer? Naz HPO4 and Na;' POA HSPO4 and NaHzE PO4 HyPO4 and Naz" HPOA NaH,PO4 and

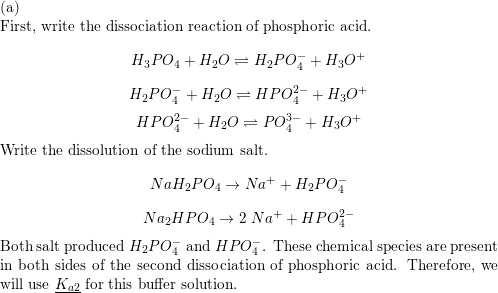





![PDF] Speciation Diagrams in Some Metal Ions-H3PO4 System | Semantic Scholar PDF] Speciation Diagrams in Some Metal Ions-H3PO4 System | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7b7664a40b485208d319b07d7e0a355c3a98b73b/12-Table3-1.png)